Drug-user advocates fear the ACT's courts may be encouraging heroin addicts to seek a largely untested and potentially dangerous implant to curb their habit.
Subscribe now for unlimited access.
$0/
(min cost $0)
or signup to continue reading
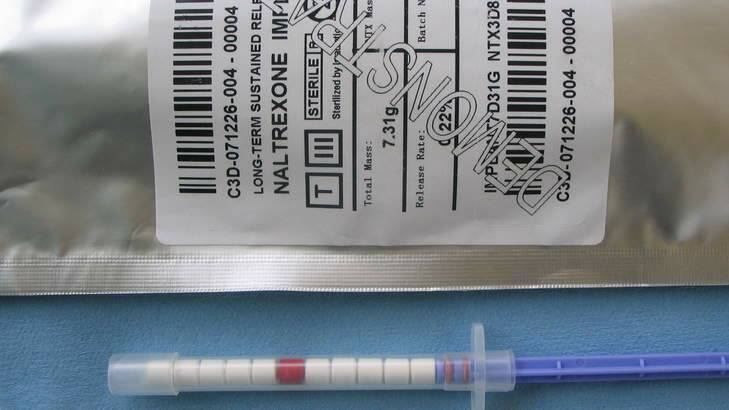
Naltrexone implants are sold as a ''miracle cure'' for opioid addiction, working by preventing the patient's brain from feeling any euphoric effect from the substances.
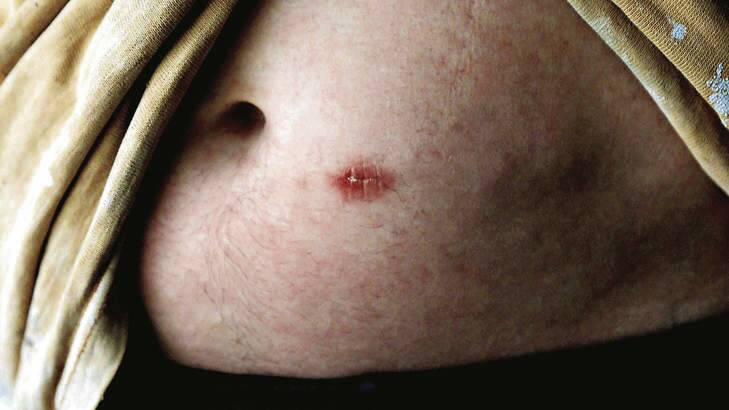
The implants have been sold outside the ACT since the early 2000s, but are not approved by the Therapeutic Goods Administration due to a lack of clinical trials.
Last September the NSW Coroner published a damning criticism of the misuse of the ''highly expensive'' implants after they were linked to the deaths of three patients at a Sydney addiction clinic named Psych n Soul.
The Australian National Council on Drugs has issued a warning, saying the implants have not been properly tested and could cause ''unknown harms, for an unknown benefit''.
Heroin-users advocacy group Canberra Alliance for Harm Minimisation and Advocacy has expressed concerns that ACT courts may be directly and indirectly condoning the use of naltrexone implants for criminals seeking rehabilitation.
In one recent example, a man being sentenced for conspiring to rob a supermarket in Canberra's south told the Supreme Court of his plans to travel interstate to get a naltrexone implant. The man, who cannot be named, told the court earlier this year that the implant would help end his lengthy battle with heroin, which had been a major factor in his offending.
Efforts to curb addiction are often taken into account during the sentencing of defendants, but the alliance warns that that naltrexone implants should not be considered a positive step for drug users. The group is working with several drug and alcohol advisory committees to develop a position paper to advise the courts and
other key agencies about the potential dangers of the implant.
''One of the reasons we wanted to prepare an ACT statement was to actually give it to the courts,'' alliance manager Nicole Wiggins said. ''We have heard of people being told … that they have to get a naltrexone implant.''
Naltrexone has been used in tablet form for more than 20 years in Australia, and with TGA approval since 1998. But less than 10 per cent of patients stick with the tablets, which are taken daily, for six months or more.
The slow-release implants were designed as a permanent fix to alleviate the need for daily dosing. They are not manufactured or sold in the ACT, and the ACT government's Alcohol and Other Drug Services refuses to refer patients for implants, citing the lack of TGA approval.
ACT Health also says it has no plan to fund the treatment.
ACT Law Society president Noor Blumer said it was appropriate for the courts to take into account any attempt by a defendant to rehabilitate in the sentencing process. Ms Blumer said the courts relied on the advice of medical practitioners, as well as reports from professional bodies, to determine what forms of rehabilitation were appropriate and effective.

