A new research project testing new drugs on the deadliest form of childhood brain cancer has been given the green light, in what could lead to major breakthroughs in treatment options for sufferers.
Subscribe now for unlimited access.
$0/
(min cost $0)
or signup to continue reading
Researchers at the Children's Cancer Institute plan to test potential new drug treatments on diffuse intrinsic pontine glioma (DIPG) tumour models to see if they are effective in shrinking tumours.
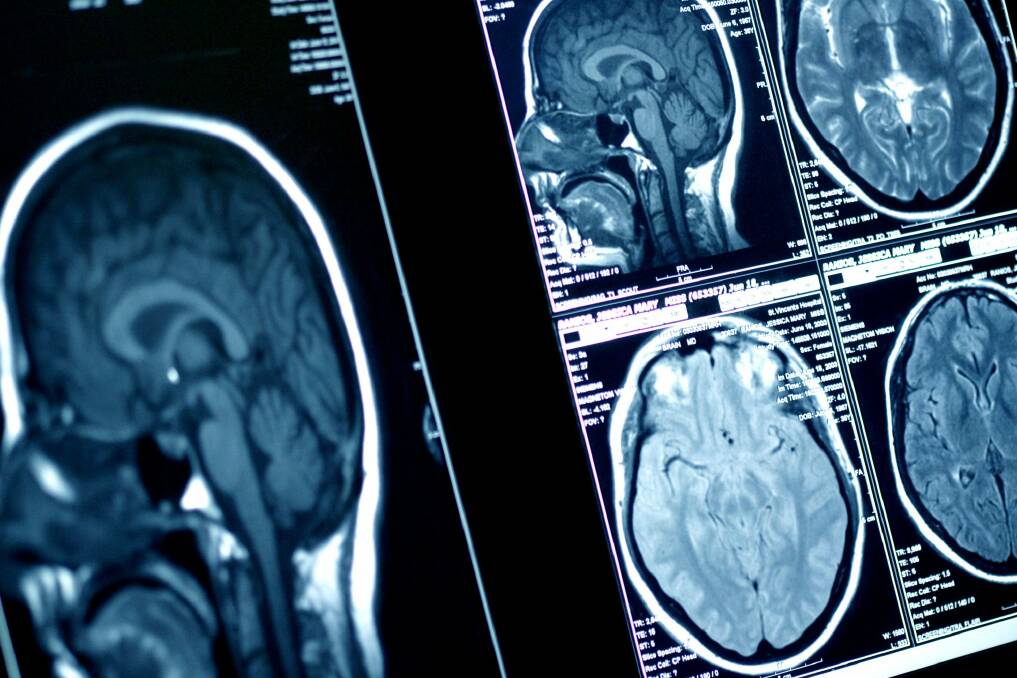
DIPG is one of the most deadly paediatric brain cancers.
Cure Brain Cancer Foundation's head of research strategy, Michelle Stewart, said DIPG was particularly difficult to treat because tumours were located within the brainstem, making it often impossible to operate, and radiotherapy was often the only treatment that could prolong survival.
Ms Stewart said radiotherapy however was often associated with severe side-effects and most children died within a year.
"Recently, the good news is, there have been advances in the way DIPG is researched and those advances mean that we are a lot closer to finding drugs that might be effective," she said.
"For the first time, there is hope for patients with DIPG. This is a massive improvement in hope and potentially survival for these patients.
"This will mean it will leapfrog the traditional research pathway because these drugs are already in use in other diseases and cancers and hopefully speed something up so we could actually have some treatment options for DIPG patients."
Brain cancer kills more Australian children than any other disease, according to Cure Brain Cancer Foundation. Only two in 10 people diagnosed with brain cancer will survive for at least five years.
Ms Stewart said it was hoped the new research could see results in about three years.
"Once those results are firmed up, we think it'll be pretty quick before they get it into patients and they might even be able to do it faster than that," she said.
The research project is expected to cost about $90,000 and has been co-funded by Cure Brain Cancer and the Campbell Edwards Trust.

