The battery has had a lot of development time put into it since Italian physicist and chemist Alessandro Volta invented the voltaic pile and published his work in 1799.
Subscribe now for unlimited access.
$0/
(min cost $0)
or signup to continue reading
One major advance was the first one to be rechargeable. That was the lead-acid battery invented in 1859 by French physicist Gaston Planté. We still use these to start most internal combustion engines today.
Most battery electric vehicles (BEVs) to date have used a lithium-based battery, but that looks like changing as well.
Advances in lithium have included replacing some of the carbon-based negative electrode with a small percentage of silicon to increase the density and therefore the range (or reduce the weight for a given capacity).
The drawback there however is the more silicon is added, the more the cell wants to expand, and that's dangerous.
Pushing lithium battery capacity too far is how you get issues like the Galaxy Note 7 phone bursting into flames.
Lithium ion also has other drawbacks though, such as scarcity of some of the materials required to keep it stable. Lithium iron phosphate, used in BYDs, Teslas and some other BEVs now, is a cobalt-free alternative, which does at least sidestep the issue of young children suffering very poor conditions and even death in unregulated cobalt mines in the Democratic Republic of Congo.
An advantage of lithium batteries is a useful range for a lot of BEVs.
A disadvantage is how astoundingly difficult lithium's electrolyte is to extinguish in the rare cases where the vehicle does catch fire. CAMS even banned EVs from competing in amateur events because clubs just don't have the capacity to deal with such a fire.
Another limitation of lithium is their available capacity (vehicle range) is greatly reduced if they get too hot (above about 45 degrees C) or too cold (below 0 degrees C).
However, there is an alternative on the horizon and that is sodium-ion.
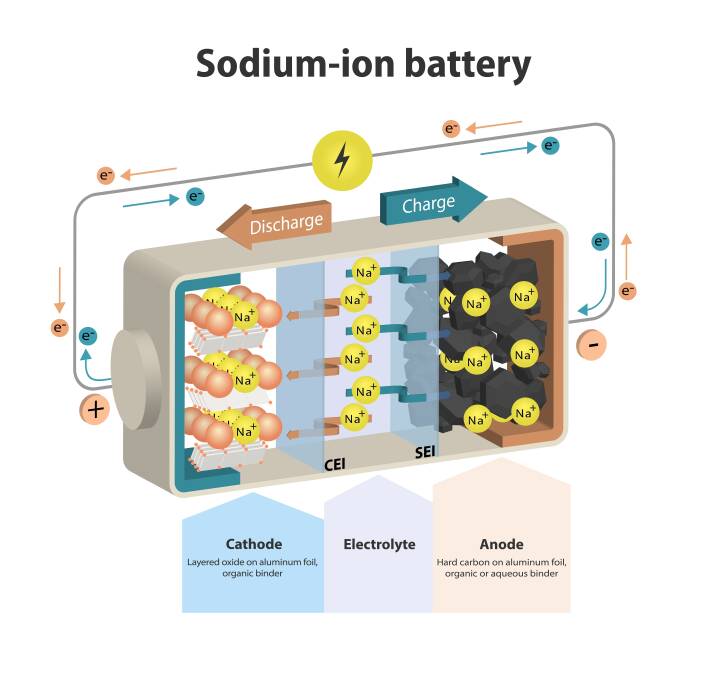
To make a sodium-ion battery, the battery's cathode gets sodium ions instead of lithium ions, and the electrolyte will be sodium salts instead of lithium salts.
Sodium ion batteries don't need cobalt, nickel, or copper either. Cheaper and more abundant raw materials should mean they're cheaper to produce as well.
Everything is a trade-off though, and one drawback is sodium's lower energy density than lithium, and so the vehicle will have a shorter range for a given physical battery size. But that's when comparing them at 20-25 degrees C, because the sodium battery has a much wider operating range making them far more suitable for desert or snow.
Sodium's density is still substantially higher than lead acid, and certainly suitable for commuter applications. Meanwhile the size and weight of a battery are irrelevant for most static storage uses.
Another advantage is safety, with development having gone into creating a non-flammable electrolyte for the sodium battery.
Battery degradation has so far been the thing holding sodium ion batteries back, and so this has been one major area of recent research and development, with some labs claiming they're reaching parity with lithium.
That's not to say lithium is good enough itself though. The average age of registered passenger vehicles in Australia is about 10 years old, which - since some die early due to damage or other causes - means the majority of them must surely be making it past 20 years before they're retired or scrapped.
Unless we want to further increase the environmental damage that would be caused by scrapping cars earlier, a lithium BEV that's been used regularly would likely need a new battery once in its life. Of course, some will start with a large capacity and be looked after by never fully charging or discharging them. Others, like fleet hacks, won't.
And I feel that here is where I should also point out that a major part of the environmental cost of making a BEV is in the manufacture (and eventually, recycling) of the lithium battery, which the sodium battery could partly, but not fully, mitigate.
Additionally, sodium batteries being cheaper to buy as replacements (if they're actually offered, that is) will likely matter to the used market from the 2030s onwards.

